
Introduction
Burrs are microscopic projections of excess material left after machining—but in medical device manufacturing, even a sub-millimeter burr can cause serious harm. Unlike general industrial parts, burrs on surgical instruments, implants, or fluid-pathway components cause direct patient harm, compromise sterility, and trigger regulatory non-compliance.
The consequences are real: FDA enforcement actions, failed sterilization validation, device recalls, and patient injury liability. In December 2024, Phillips Precision Inc. received an FDA warning letter citing unvalidated deburring processes and leftover burrs on orthopedic implants.
Medical device recalls reached 1,059 events in 2024—a four-year high—with device failures accounting for 11.1% of all recalls.
This guide covers the why, how, and what of medical device deburring: removal methods, detection techniques, compliance standards (ISO 13485, FDA 21 CFR Part 820, ISO 10993), and the documentation practices that hold up under audit.
TL;DR
- Burrs represent patient safety hazards and regulatory compliance risks—not cosmetic defects
- Choosing the wrong removal method for your device type, material, or regulatory class risks part rejection and costly rework
- Surface finish directly affects sterilization efficacy and biocompatibility testing
- ISO 13485 requires validated deburring processes with measurable acceptance criteria—and FDA auditors will scrutinize your documentation just as closely as your parts
Why Burr Removal Is Critical in Medical Device Manufacturing
Burrs are raised, sharp projections left after machining operations—CNC milling, drilling, turning, or stamping. In medical applications, even a burr measuring a few hundredths of a millimeter can cause serious harm.
Patient Safety Impact:
- Tissue damage: Burrs on implants abrade tissue during normal movement
- Metallic particle release: Burrs on fluid pathway components shed particles into the bloodstream or drug delivery systems
- Surgical injury risk: Sharp edges on instruments create handling hazards for surgeons and sterile processing staff
The FDA documented that metal-on-metal hip implants release tiny particles causing Adverse Local Tissue Reaction (ALTR). Approximately one million Americans face premature revision surgery due to metallosis, with DePuy's ASR settlement costs exceeding $4 billion.
Sterilization and Bioburden Risk:
Surface roughness caused by unremoved burrs directly undermines sterilization effectiveness. Rough, burred surfaces harbor bacteria, endotoxins, and organic residue that standard autoclave or EtO cycles cannot reliably eliminate. Research shows that autoclaving Ti6Al4V increases biofilm-related gene expression 4-9 times compared to UV sterilization, linking surface treatment choices directly to infection risk.
Biocompatibility Failures:
ISO 10993 biocompatibility testing flags elevated cytotoxicity or irritation responses when machined surfaces carry burrs or excessive roughness. Greater surface area accelerates particle release — and can stall or block device approval outright.
Regulatory Enforcement:
FDA inspectors specifically examine whether deburring is a controlled, validated process within a manufacturer's quality management system. Without documented evidence of that control, manufacturers face 483 observations, warning letters, or product recalls.
The Phillips Precision warning letter illustrates what that scrutiny looks like in practice: violations included blades received with "leftover burrs," metal debris, and cleaning validation that failed to challenge worst-case surface finishes.
Deburring Methods for Medical Devices
No single deburring method works for all medical device applications. The correct method depends on material (titanium, stainless steel, PEEK), device geometry, required surface finish Ra value, production volume, and regulatory classification (Class I, II, or III device).

Mechanical Deburring
Manual filing, grinding, and brushing represent the baseline approach: low cost, flexible for complex geometries, but operator-dependent. This makes validation challenging under ISO 13485, limiting suitability to non-critical, lower-risk components.
CNC-driven mechanical deburring—using multi-axis machining to apply controlled edge-rounding passes—offers a more repeatable and documentable alternative. When tight geometric tolerances must be maintained alongside deburring, programmed chamfering operations produce consistent results suitable for batch validation.
Vibratory and Mass Finishing
Vibratory deburring (vibe deburring) and drag finishing place parts in a bowl or fixture with abrasive media and subject them to controlled motion. This produces uniform edge rounding and surface smoothing across complex geometries without manual contact, making it highly repeatable and suitable for ISO 13485 validation.
Key advantages:
- Batch processing capability with consistent results
- Reaches complex internal geometries
- Eliminates operator variability
- Supports process validation with measurable parameters
For high-value, delicate components—orthopedic implants, hearing aid parts—drag finishing clamps parts individually and pulls them through media, eliminating the risk of surface marks or nicks. CNC Programming Solutions' vibe deburring service applies this approach to precision medical components, with documented process parameters and auditable batch records to support ISO 13485 validation requirements.
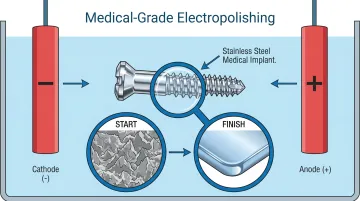
Electrochemical Deburring (Electropolishing)
Electropolishing is an electrochemical process that dissolves surface material preferentially at high points (burrs, peaks) to produce a smooth, passive, corrosion-resistant surface. Electropolishing may improve Ra values by up to 50 percent, though mechanical pre-polishing may be needed for ultra-low finishes.
ASTM F86 and ASTM B600 recommend electropolishing as the preferred surface treatment for medical implants, instruments, and invasive devices — advisory standards rather than mandated requirements, but widely expected by auditors and notified bodies.
Critical benefits for medical devices:
- Removes micro-burrs that mechanical methods miss
- Creates chromium-enriched passive layer on stainless steel (improving corrosion resistance)
- Often required for implantable or fluid-contact devices
- Must be followed by passivation per ASTM A967 for full compliance
Thermal and Laser Deburring
Thermal Energy Method (TEM) uses controlled combustion to burn away thin burrs without affecting the base material. It's particularly effective for internal channels and geometries where mechanical tools can't reach — but strict process controls are essential to avoid heat-affected zones on sensitive alloys.
Laser deburring takes a different approach: ultrafast femtosecond and picosecond pulses remove micro-burrs through ablation and vaporization rather than melting. Femtosecond lasers are already used to process nitinol, cobalt-chromium, and platinum-iridium for cardiovascular stents. Wet/submerged laser cutting further improves results by eliminating dross, making this method increasingly common for Class III device manufacturing.
How to Detect Burrs and Assess Deburring Quality
Manual visual and tactile inspection is subjective, inconsistent, and insufficient for small internal burrs or features below a few hundredths of a millimeter. This makes it inadequate as a standalone quality control method under ISO 13485's validation requirements.
More reliable alternatives exist. Coordinate Measuring Machines (CMMs) scan complex curved surfaces and flag dimensional deviations caused by burr presence, while optical and laser surface profilometers quantify roughness using standardized parameters.
Ra Benchmarks by Device Type:
| Device/Component | Ra Specification | Application |
|---|---|---|
| Femoral heads | 0.010-0.015 μm | Articulating surface |
| Hard-on-hard bearings | < 0.025 μm | Ceramic/CoCr joints |
| Surgical instruments | 0.4-0.8 μm | General specification |
| Instrument functional surfaces | < 0.2 μm | Cutting edges, gripping |
| Cardiac/arterial fluid pathways | < 100 nm Sa | Prevent thrombosis |
| Osseointegration zones | 1.0-2.0 μm Sa | Bone-contact surfaces |
Source: Criterion Precision, Surface Finish Standards
These targets vary by nearly two orders of magnitude, so confirming the correct Ra specification before any deburring process begins is essential to avoid over- or under-finishing.
Validation Best Practices
A compliant post-deburring validation process should include:
- Surface roughness measurement against the device-specific Ra specification
- Visual inspection under magnification (SEM or stereo microscope for critical components)
- Periodic cross-section analysis for internal features and blind holes
- Documented results tied to production batch records for ISO 13485 traceability

Compliance Standards That Govern Medical Device Deburring
ISO 13485 Requirements
ISO 13485 requires manufacturers to:
- Define surface finish as a critical design control attribute
- Validate all finishing processes (including deburring) to demonstrate consistent, repeatable results
- Maintain documented procedures and records
"We deburr all parts" is not sufficient. A validated, controlled process with acceptance criteria is required.
Clause 7.5.6 - Special Process Validation:
ISO 13485 Clause 7.5.6 classifies deburring as a special process when output cannot be fully verified by inspection alone. Validation must include Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Internal channel deburring and electropolishing both require special process validation.
FDA 21 CFR Part 820 (QMSR)
The FDA's Quality Management System Regulation became effective February 2, 2026, incorporating ISO 13485:2016 by reference and eliminating previous record review exemptions.
Device History Record (DHR) Requirements:
21 CFR 820.184 requires acceptance records demonstrating that deburring and surface finishing were performed per approved procedures, with inspection results. Every production batch must document:
- Dates of manufacture
- Quantity manufactured and released
- Acceptance records per Device Master Record specification
- Inspection/measurement results
ISO 10993 (Biocompatibility Evaluation)
Surface finish is a key variable in biocompatibility testing because rougher surfaces increase particle release and surface area in contact with tissue. ISO 10993-18:2020 requires chemical characterization on the final, finished medical device, ensuring extractables profiles include impacts from actual manufacturing and sterilization methods.
Deburring and finishing processes must be performed on test samples that represent the final device surface. In practice, this means the post-deburring surface state must be locked in before biocompatibility testing is completed.
ASTM F86 (Surface Preparation of Metallic Surgical Implants)
Surface chemistry requirements go further for implantable devices. ASTM F86-21 covers implants manufactured from iron, cobalt, nickel, titanium, and tantalum base materials. The standard specifies chemical and electrochemical surface treatments to remove contaminants — iron particles, ceramic media, and foreign particles — embedded during forming, machining, tumbling, or media blasting. The objective is to improve corrosion resistance and establish a passive oxide film on the metal surface.
Documentation Requirements for Audit Readiness
Manufacturers should maintain:
- Validated SOPs with defined process parameters
- Equipment calibration records tied to measurement instruments
- Inspection/measurement results per batch linked to DHR
- Nonconformance records for burr-related rejections
- Corrective action documentation when defects occur

Auditors look for evidence that deburring is systematic, not reactive. The Phillips Precision warning letter illustrates the failure mode: validation that does not challenge worst-case product geometries or surface finishes is inadequate.
Conclusion
Burr removal in medical device manufacturing is a non-negotiable quality and compliance function—not a finishing afterthought. The right deburring method, properly validated and documented, directly protects patient safety, supports sterilization validation, and keeps the manufacturer inspection-ready.
Manufacturers who get this right focus on three things:
- Align method selection with device classification and material requirements
- Establish validated processes with measurable acceptance criteria (Ra values, edge condition)
- Maintain complete documentation to satisfy FDA, ISO 13485, and ASTM requirements
Partnering with experienced finishing service providers can accelerate compliance without slowing production. For companies seeking vibratory deburring capabilities, CNC Programming Solutions offers vibe deburring services built to produce consistent, auditable surface results for precision component manufacturing.
Frequently Asked Questions
What is medical device compliance?
Medical device compliance refers to a manufacturer's adherence to applicable regulatory standards and quality system requirements—including FDA regulations (21 CFR Part 820), ISO 13485, and device-specific standards—that govern design, manufacturing, testing, and post-market surveillance to ensure device safety and effectiveness.
What is the difference between ISO 13485 and IEC 60601?
ISO 13485 is a quality management system standard covering the entire medical device manufacturing process (including manufacturing controls like deburring), while IEC 60601 is a safety and performance standard specifically for electrical medical equipment. Manufacturers of electrical devices often need to satisfy both, but they serve distinct compliance functions.
What deburring method is best for implantable medical devices?
Electropolishing combined with passivation is the standard choice for implantable devices because it removes micro-burrs, improves corrosion resistance, and produces a biocompatible surface. Drag finishing or vibratory deburring may be used for pre-polishing, and the final method must be validated per ISO 13485.
Can burrs on medical devices cause regulatory failures or product recalls?
Yes—burrs and inadequate surface finishing are recognized causes of medical device nonconformances. They can result in FDA 483 observations during inspections, and in severe cases where patient harm or sterility failures occur, they can trigger recalls and FDA warning letters.
What Ra surface finish value is typically required for medical device compliance?
Ra requirements vary by device type: implantable devices typically require Ra 0.2–0.4 μm, surgical instruments Ra 0.4–0.8 μm, and fluid pathway components Ra below 0.1 μm. Manufacturers must define and document the required Ra value as part of their design controls under ISO 13485.
How should deburring processes be documented for FDA audits?
Manufacturers should maintain:
- Validated SOPs for each deburring method
- Records of process parameters and equipment calibration
- Per-batch inspection results tied to the device history record (DHR)
- Nonconformance and corrective action records related to surface finish
FDA inspectors review these documents to confirm deburring is a controlled, repeatable process.


