
Introduction
In medical device manufacturing, surface finish directly determines patient safety, biocompatibility, sterilizability, and regulatory approval outcomes. Yet many manufacturers face the same specification challenge: there is no single universal finish requirement. The correct spec depends on device type, functional zone, material, and applicable standards—and getting it wrong can mean device failure, infection risk, or regulatory rejection.
A single implant illustrates the complexity: it may need an ultra-smooth articulating surface at Ra < 0.05 µm to prevent wear debris, a moderately rough bone-contact zone at Sa 1–2 µm to promote osseointegration, and a cleanable external surface at Ra 0.4–0.8 µm, all on the same component.
TLDR
- Surface finish is measured by Ra (arithmetic mean roughness), Rz (peak-to-valley height), and Sa (areal height)—not just general "smoothness"
- Articulating implant surfaces need ultra-smooth finishes (Ra < 0.05 µm), bone-contact zones require controlled roughness (Sa 1–2 µm), and surgical instruments typically target Ra 0.4–0.8 µm for cleanability
- ISO 4287/25178, ASTM F86/F136, and FDA 21 CFR Part 820 form the core regulatory framework for specification, measurement, and documentation
- Ra alone is insufficient—pair it with Rz, Sa, or Sdr depending on the functional application to avoid over- or under-specifying critical surfaces
What Is Surface Finish in Medical Devices?
Surface finish refers to the microscopic texture and topography of a component's surface, measured in micrometers (µm). In medical devices, it functions as a design parameter, not just a manufacturing output, because it governs biocompatibility, wear resistance, corrosion resistance, and cleanability.
The stakes run in both directions. A surface that is too rough may harbor bacteria or generate wear debris; one that is over-polished may prevent bone integration or coating adhesion.
Key Surface Roughness Parameters
Ra (arithmetic mean roughness) is the most common parameter, averaging surface deviations from the mean line. The problem: Ra cannot distinguish between a surface with dangerous high peaks and one with deep lubrication-retaining valleys. According to NIST's Surface Finish Metrology Tutorial, Ra loses sign information and cannot detect isolated asperities or valley features critical to function or contamination risk.
Complementary parameters:
- Rz (maximum peak-to-valley height) — captures extreme peaks and valleys Ra misses; defined in ISO 4287 for 2D profile measurement
- Sa and Sz — 3D areal equivalents per ISO 25178-2:2021; essential for complex implant geometries where a single profile line misses critical features
- Sdr (developed interfacial area ratio) — measures the additional surface area created by texture, making it the key metric for osseointegration surfaces

Why "Smoother Is Not Always Better"
Choosing the right parameter set is only half the challenge — knowing what finish value to target is the other. Over-polished surfaces can reduce coating adhesion, impair osseointegration, and increase friction in some lubrication regimes. The optimal finish must match the functional zone, not default to the finest achievable finish.
Dental implants illustrate this clearly: bone-contact zones deliberately require moderately rough surfaces (Sa 1–2 µm) to promote bone cell anchoring, while the abutment interface must stay smooth to resist bacterial accumulation.
Surface Finish Requirements by Medical Device Type
A single device may require multiple different finishes applied to different functional zones—articulating, bone-contact, soft-tissue, and non-contact surfaces all carry different specification logic.
Implantable Devices
Articulating surfaces (hip/knee femoral heads, hard-on-hard bearings):
Ultra-smooth finishes are essential here — even small deviations generate wear debris that drives osteolysis and implant failure. Industry practice typically targets Ra 0.01–0.05 µm for femoral heads and hard-on-hard bearings, though ASTM F2033 does not publicly mandate specific Ra values.
Research on wear debris characterization shows that higher counterface roughness increases asperity interactions, wear rates, and particle generation. These particles trigger macrophage activation and pro-inflammatory mediators (TNF-α, RANKL, IL-1β), contributing to osteolysis and aseptic loosening.
Cobalt-chromium alloys are preferred for achieving these ultra-fine finishes due to superior hardness (approximately 797 HV vs. 645 HV for Ti-6Al-4V) and their ability to maintain mirror polishes in service.
Osseointegration zones (dental implants, cementless orthopedic stems):
Moderately rough surfaces are intentionally required to promote bone cell anchoring. The Wennerberg and Albrektsson classification, widely cited in implant dentistry, categorizes areal roughness as:
- Smooth: Sa < 0.5 µm
- Minimally rough: Sa 0.5–1.0 µm
- Moderately rough: Sa 1–2 µm (optimal for osseointegration)
- Rough: Sa > 2 µm
Systematic reviews confirm that moderately rough surfaces are associated with favorable osseointegration and secondary stability. The distinction is critical: smooth abutment surfaces (typically Ra < 0.2 µm) resist bacterial accumulation, while bone-contact zones require controlled roughness for cell attachment.

Non-articulating/non-contact surfaces:
General implant surfaces not involved in articulation or bone contact accept a broader Ra range of 0.4–1.6 µm. However, biocompatibility and corrosion resistance still govern minimum requirements.
Surgical Instruments
Where implantable devices prioritize biological response, surgical instruments face a different challenge: reusable surfaces must survive hundreds of sterilization cycles while remaining cleanable. Industry consensus holds that Ra 0.8 µm is the critical threshold above which organic soil and microbial load retention increases significantly, making the Ra 0.4–0.8 µm range the standard target (though this threshold does not appear as a mandate in freely accessible ISO 17664 or AAMI standards).
Key considerations:
- Functional surfaces (cutting edges, articulating joints on scissors/forceps) require tighter finishes than general surfaces
- Electropolishing is preferred over mechanical polishing alone—it reduces surface roughness while building a passive oxide layer that resists autoclave-induced corrosion (a mechanism confirmed in ASTM F86-21, which describes chemical and electrochemical treatments for implant surfaces)
Cardiac and Fluid-Flow Devices
Devices in contact with blood flow—stents, heart valves, arterial grafts—require extremely smooth surfaces, often sub-100 nm Sa. Proteins initiating clotting respond to fine-scale surface texture, and rougher surfaces promote thrombosis. Research on blood-material interactions shows that micrometer-scale roughness increases available surface area and protein adsorption; nanoscale features can further increase fibrinogen adsorption and modulate platelet activation.
Smooth surfaces also reduce hemolysis from blood cell damage at constrictions. A 2026 study on blood pump surfaces found that rougher titanium alloy surfaces increased hemolysis rates — confirming that machining parameters directly affect hemocompatibility outcomes.
Manufacturing processes can reliably hit these targets: electropolishing of NiTi stent tubing has achieved Ra ≈ 46.9 nm, demonstrating what's achievable with optimized finishing.
Key Standards Governing Medical Device Surface Finish
Surface finish compliance involves three distinct layers: measurement standards (ISO), material standards (ASTM), and regulatory quality requirements (FDA). Each operates independently, but all three must appear in your device documentation.
ISO Standards for Surface Measurement
ISO 4287 defines 2D profile parameters (Ra, Rz, Rq, RSm) measured along a single line. ISO 4288:1996 governs assessment rules and cutoff selection.
ISO 25178-2:2021 extends these to 3D areal parameters (Sa, Sz, Sdr) that capture complex surface features missed by profile measurement—critical for dental implants, porous coatings, and curved geometries.
ISO 1302 (now replaced by ISO 21920-1:2021) governs how surface texture is indicated on engineering drawings, ensuring consistent communication between design and manufacturing.
ASTM Standards for Medical Device Materials
Two standards frame material and surface preparation requirements, but neither prescribes Ra values directly:
- ASTM F136 — Specifies wrought Ti-6Al-4V ELI alloy for surgical implants (chemical, mechanical, and metallurgical requirements). Ra limits must be defined in engineering drawings based on intended use.
- ASTM F86-21 — Covers surface preparation and marking of metallic implants, including passivation to remove contaminants and form a protective oxide film. Roughness limits remain the manufacturer's responsibility.
FDA Regulatory Documentation Requirements
Material and measurement standards define what to achieve — FDA regulation defines how to document it. The FDA finalized the Quality Management System Regulation (QMSR) on February 2, 2024, incorporating ISO 13485:2016 by reference, with a compliance date of February 2, 2026.
Surface finish must be addressed as:
- Design Input (ISO 13485 Clause 7.3) when it affects safety and performance
- Validated process (Clause 7.5.6) for finishing processes like passivation and electropolishing, because their outcomes cannot be verified by non-destructive inspection alone
- Calibrated measurement (Clause 7.6) with traceability to national standards (NIST or equivalent)
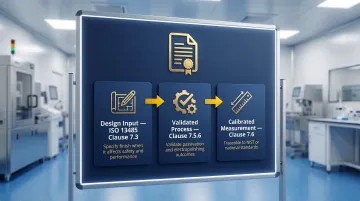
Changes to surface finish specifications may require regulatory notification depending on device classification.
How to Specify, Measure, and Validate Surface Finish Correctly
Compliance depends on three connected steps: clear specification, accurate measurement, and documented validation. A gap in any one of them creates direct regulatory exposure — and the most common gaps are in specification and post-processing validation.
Specification Best Practices
Applying a single finish requirement across an entire component is a common specification error. Zone-specific requirements are the correct approach:
- Use ISO 1302/21920-1 symbols on engineering drawings
- Specify parameter (Ra, Rz, Sa), upper/lower limits, measurement orientation, and evaluation length together
- Define whether roughness applies pre- or post-treatment (passivation, electropolishing, and sterilization all alter surface characteristics)
- Validate surface in its final, as-used state
Measurement Methods
Contact profilometry (stylus-based 2D measurement) traces surface profiles with a diamond stylus to measure Ra/Rz. Key setup variables affect the result:
- Stylus tip radius - larger tips cannot follow narrow valleys, skewing Ra/Rz readings lower on sharp textures
- Cutoff length (λc) - separates roughness from waviness; ISO 4288 provides rules for cutoff selection based on Ra range and surface type
Two inspectors using different cutoff lengths can measure significantly different Ra on the same surface, per NIST guidance.
These setup dependencies make measurement methodology a specification decision, not just a shop floor choice. Non-contact optical methods — white light interferometry, confocal microscopy — handle 3D areal measurement (Sa/Sz) per ISO 25178-600:2019 and are preferred for:
- Complex geometries where stylus access is limited
- Delicate surfaces where contact may cause damage
- Soft materials
- Comprehensive surface mapping beyond single-line profiles
Instrument vertical resolution must match the tolerance being measured. Calibration must be traceable to national standards under ISO 13485 Clause 7.6.
Process Validation and Control
Initial process validation demonstrates consistent finish achievement across the tolerance range. Statistical process control provides ongoing monitoring. FDA guidance does not prescribe numeric Cpk thresholds, but industry practice commonly targets Cpk ≥ 1.33 for critical surfaces and Cpk ≥ 1.67 for ultra-critical surfaces.
Surface finish must be validated after all post-processing steps — not before. ASTM F86-21 confirms that passivation and other chemical/electrochemical treatments alter surface characteristics. Some materials also change measurably across sterilization cycles, which means validation on pre-sterilized parts can produce results that don't reflect the device's actual in-use condition.
Common Surface Finish Specification Mistakes
Relying on Ra Alone
Ra is an average that cannot detect dangerous isolated peaks (which drive abrasive wear) or deep valleys (which affect lubrication retention and cleanability). For articulating surfaces, pair Ra with Rz or peak parameters (Rpk). For osseointegration zones, add Sa and Sdr. For complex geometries, use 3D areal measurement per ISO 25178.
Over-Specifying Non-Functional Surfaces
Demanding ultra-smooth finishes on handles, external housings, or non-contact areas drives cost exponentially through multiple manual polishing steps without adding clinical value. Specifications should be tied to function, not habit or aesthetics. A non-contact surface at Ra 1.6 µm may be entirely adequate where Ra 0.05 µm is unnecessary.
Ignoring Post-Processing Interactions
Finish specifications must state whether roughness applies pre- or post-treatment. Passivation, electropolishing, and repeated sterilization cycles all alter surface characteristics. Research on magnesium alloys shows that sterilization can measurably shift surface roughness values — particularly in reactive alloys. Validate the surface in its final, as-used state to ensure the specification holds through the product lifecycle.
How CNC Programming Solutions Can Help
CNC Programming Solutions offers precision machining and finishing services directly applicable to achieving the surface finish targets medical device components require. Based in Perkins, Oklahoma, the company operates CNC capabilities up to 5-axis milling—ideal for complex medical geometries including medical implants—paired with in-house finishing services:
- Bead blasting removes imperfections and prepares surfaces for subsequent finishing
- Vibratory deburring removes sharp edges and burrs that can trap contaminants or cause tissue irritation
- Anodizing creates protective oxide layers that enhance corrosion resistance and biocompatibility

These processes address both functional requirements (cleanability, osseointegration preparation) and regulatory needs (biocompatibility, corrosion resistance).
Achieving tight surface finish targets on complex medical geometries starts with the machining program itself. CNC Programming Solutions' expertise in tool path strategy, cutting parameters, and tool selection determines the as-machined Ra — before any secondary finishing begins.
Working with a machining partner who understands the full finishing sequence reduces secondary operations, shortens lead times, and lowers the risk of surface finish non-conformances at inspection.
For medical device manufacturers managing zone-specific finish requirements, tight tolerances (±0.001 inches / ±0.025 mm for high-precision parts), and multi-step finishing sequences, a single partner who integrates programming, machining, and finishing shortens the path from design to validated production.
Contact CNC Programming Solutions:
- Email: cncsolutions22@gmail.com
- Phone: 405-714-3714
- Address: 2322 W. 104th Street, Perkins, OK 74059
Conclusion
Selecting the right surface finish for a medical device is a zone-by-zone engineering process tied to functional requirements, material properties, and regulatory obligations — and there is no universal specification. Articulating surfaces demand ultra-smooth finishes to prevent wear debris, osseointegration zones require controlled roughness to promote bone anchoring, and cleanable surgical instruments target an intermediate range for sterilizability.
Each zone must be specified with the appropriate parameters (Ra paired with Rz, Sa, or Sdr), measured with calibrated instruments, and validated through process controls that account for post-processing treatments.
Treat surface finish as a living specification: revisit it at every design iteration and after any change to materials, manufacturing processes, or post-processing steps. Even minor process shifts can push surface texture outside validated ranges and introduce compliance risk.
Frequently Asked Questions
What is the standard surface finish for machined parts?
Ra 3.2 µm (125 µin) is the commonly accepted default for general CNC machined surfaces. However, medical device components typically require much finer finishes—the correct target depends on device type, functional zone, and applicable standards rather than a universal default.
What is Ra 3.2 surface finish?
Ra 3.2 µm is the arithmetic mean roughness produced by standard CNC machining: visibly smooth but not mirror-polished. This finish is too rough for most medical device applications; articulating implant surfaces and surgical instruments require Ra values from 0.8 µm down to 0.01 µm depending on function.
What is medical device coating?
Medical device coatings are surface treatments (anodizing, parylene, hydroxyapatite, titanium plasma spray, electropolishing) applied after machining to enhance biocompatibility, corrosion resistance, osseointegration, or sterilizability. The base finish must meet specifications before coating, since coatings over rough surfaces may not adhere uniformly or satisfy regulatory requirements.
What device is used to measure surface finish?
Contact profilometers (stylus-based) are standard for 2D Ra/Rz measurement; white light interferometers and confocal microscopes handle 3D areal parameters (Sa, Sz) and delicate surfaces. All instruments must be calibrated to traceable national standards to meet ISO 13485 inspection requirements.
What surface finish is required for surgical implants?
Requirements vary by zone: articulating surfaces typically require Ra < 0.05 µm, osseointegration surfaces target Sa 1–2 µm, and non-contact surfaces accept Ra 0.4–1.6 µm. A single implant may require multiple different finishes applied to different functional areas based on wear, bone integration, and cleanability needs.
Is Ra alone sufficient to specify a medical device surface finish?
No. Ra alone is generally insufficient because it is an average that cannot distinguish between surfaces with dangerous isolated peaks or beneficial deep valleys. Pair Ra with Rz for wear-critical surfaces, Sa and Sdr for osseointegration zones, and use 3D areal parameters for complex geometries where a single profile line misses critical surface features.


