
Introduction
Yes, brass can be anodized — but there are real caveats most surface finishing guides skip. Alloy composition and dimensional tolerances directly determine whether the process succeeds or produces costly rework.
Brass anodizing differs fundamentally from aluminum anodizing. Aluminum forms a uniform, predictable oxide layer governed by established industry specifications. Brass, by contrast, produces a mixed copper-zinc oxide layer with variable chemistry and inconsistent growth behavior.
Engineers who carry aluminum-derived expectations into brass anodizing projects frequently encounter patchy finishes, unexpected dimensional changes, and adhesion failures.
This guide is written for engineers, machinists, and product designers specifying surface finishes for brass components — covering the electrochemistry, quality controls, and the situations where alternative finishes make more sense.
TL;DR
- Brass can be anodized, but alloy matters — alpha brasses (up to ~35% zinc) respond consistently; high-zinc alloys produce patchy, unreliable results
- The resulting oxide layer (CuO, Cu₂O, ZnO) is mixed rather than uniform, which explains why brass anodizing is less predictable than aluminum
- Key benefits include measurable corrosion resistance (up to 2480 ohm-cm² in testing) and decorative color options
- No MIL-SPEC or ASTM standard governs brass anodizing, unlike aluminum's well-documented Type I/II/III classifications
- When documented performance standards matter, electroless nickel or hard chrome plating on brass is the more reliable choice
What Is Brass Anodizing?
Brass anodizing is an electrochemical surface treatment that converts the outer layer of a brass component into a stable oxide coating through controlled oxidation in an electrolytic bath. Unlike plating (which deposits external material), anodizing grows the oxide layer from the brass substrate itself, creating a chemically bonded surface that improves corrosion resistance, hardness, and enables color customization while retaining the part's metallic character.
The result is a harder, more corrosion-resistant surface with optional decorative color — without adding significant weight or altering the component's geometry beyond micron-scale dimensional changes.
How It Differs from Aluminum Anodizing
Aluminum anodizing produces pure aluminum oxide (Al₂O₃) — a uniform compound with predictable pore structure that enables standardized thickness specs and documented dimensional growth ratios.
Brass is different. Research published in the Materials journal confirms that brass anodizing produces a composite oxide layer containing multiple copper and zinc species: CuO, Cu₂O, Cu(OH)₂, ZnO, and Zn(OH)₂.
This mixed-species chemistry creates several practical differences:
- Aluminum anodizing follows Type I, II, and III classifications with defined layer depths; brass has no equivalent standard
- Color consistency varies because the Cu:Zn ratio in the oxide layer shifts with applied voltage
- Aluminum follows established dimensional growth ratios (33% buildup / 67% penetration for Type II); brass offers no such predictable rule

MIL-PRF-8625F, the governing specification for aluminum anodizing, explicitly warns that brass is a "non-aluminum part" that "would be attacked by pretreatment or anodizing solutions," reinforcing that the two processes cannot be treated as interchangeable.
How the Brass Anodizing Process Works
The brass anodizing workflow follows seven phases: surface preparation, solution preparation, solution application, electrochemical anodizing, optional dyeing, sealing, and quality inspection. Each stage directly affects adhesion quality, layer thickness, and color uniformity.
Required Inputs
You'll need four core inputs before starting:
- Clean brass substrate (free of oils, oxides, and machining residues)
- Electrolyte solution — phosphoric acid at 1:10 ratio with distilled water for industrial applications, or 0.1 M NaOH for research use
- Non-metallic container (to prevent contamination)
- Controlled DC power source capable of maintaining steady voltage and current density throughout the run
Core Electrochemical Transformation
During anodizing, the brass component acts as the anode in an electrolytic cell. Applied current causes oxidation at the surface, converting metallic copper and zinc into their respective oxides. This oxide layer grows both into the metal substrate and above the original surface — a critical dimensional consideration for precision parts with tight tolerances or threaded features.
Process control variables determine outcome quality:
- Voltage: Ranges from 1–60 V depending on desired layer properties; higher voltage increases the Cu:Zn ratio in the oxide from 2.6 at 30 V to 3.6 at 60 V
- Current density: Must be held within defined ranges for consistent layer growth
- Bath temperature: Typically room temperature (25°C); deviations affect oxidation rates
- Immersion time: Research from Afshar et al. demonstrated optimal corrosion resistance at 1 V for 60 minutes in 0.1 M NaOH
Step 1: Surface Cleaning and Preparation
Skip degreasing and the anodized layer won't adhere evenly. Oils, machining residues, or surface oxides all cause inconsistent oxidation and adhesion failure. The Copper Development Association specifies degreasing with organic solvents or inhibited chemical cleaners, explicitly advising against steel wool (which may contain rust inhibitors that stain copper alloys).
The standard preparation sequence:
- Soap and water cleaning to remove gross contamination
- Chemical degreaser application
- Warm-water rinse to remove all residues
- Drying before electrolyte contact
Surface roughness telegraphs through the anodized finish. Parts with smoother pre-anodized surfaces consistently achieve better adhesion and color uniformity.
Step 2: Anodizing Bath Setup and Solution Application
The prepared electrolyte solution goes into a non-metallic container. The brass part is either fully immersed or spray-coated, then left to soak for approximately 30 minutes — long enough for the solution to work into surface irregularities. Once the soak is complete, rinse and dry the part before moving to the electrochemical stage.
Step 3: Electrochemical Anodizing
The brass part is connected as the anode in the anodizing machine, with voltage and temperature set according to manufacturer parameters or application-specific requirements. Current flows through the electrolyte, oxidizing the brass surface and forming the bonded oxide layer.
Parameter consistency at this stage is what separates a uniform finish from a defective one. Voltage shifts alter the Cu:Zn oxide ratio and final color, temperature swings change the growth rate, and uneven current density produces inconsistent layer thickness across complex part geometries.
Step 4: Dyeing (Optional) and Sealing
Apply dye while the oxide layer remains open after anodizing. The porous structure pulls dye molecules in, creating color options well beyond brass's natural gold tone — including black, blue, green, red, brown, and custom shades.
Sealing closes the oxide pores, locking in color and improving corrosion resistance. Hot water sealing or chemical sealants are most common. Improper sealing leaves the oxide layer vulnerable to moisture penetration, dye leaching, and reduced wear resistance.
Benefits and Applications of Brass Anodizing
Core Functional Benefits
Anodized brass delivers measurable improvements in four areas:
- Corrosion resistance: Testing on yellow brass (CuZn35.5) achieved 2480 ohm-cm² total corrosion resistance and corrosion current density of 1.14 microA/cm², both outperforming bare brass
- Wear and abrasion resistance: The oxide layer (CuO, Cu2O, ZnO) is harder than base brass, though more brittle
- Electrical insulation: The oxide acts as a dielectric barrier, slightly reducing electrical conductivity
- Anodized surfaces wipe clean with mild detergent and resist tarnishing with minimal upkeep
Aesthetic Value
Natural anodized brass retains its warm gold/yellow tone from the underlying alloy. Dyeing opens a broader palette — black for industrial components requiring matte finishes, blue and green for architectural accents, red and brown for jewelry and decorative hardware. The metallic character stays visible beneath the oxide, unlike opaque powder coating, which hides the metal entirely.
Application Sectors
Anodized brass appears across a range of industries where both durability and appearance matter:
- Door handles, railings, façade elements, and window frames where outdoor exposure demands tarnish resistance
- Jewelry and decorative hardware benefiting from custom colors and extended longevity
- Marine fittings on non-critical components exposed to saltwater
- Consumer electronics trim, automotive accents, and lighting fixtures where tarnish would degrade appearance over time
- Dental tools, surgical instruments, and orthodontic brackets requiring corrosion resistance and easy sterilization
For applications like signage, logo plaques, and metal tags, electrochemical coloring — a related but distinct process — offers tighter control over the final aesthetic finish.
Key Factors That Affect Brass Anodizing Quality
Brass Alloy Composition
The copper-to-zinc ratio is the primary determinant of anodizing consistency. Research from Wieland-Chase establishes approximately 35% zinc as the critical threshold separating reliable from problematic anodizing behavior.
| Brass Type | Zinc Content | Phase Structure | Anodizing Behavior |
|---|---|---|---|
| Alpha brass (C260, C270) | Up to ~35% Zn | Single-phase FCC | Uniform oxidation; inhibitors (As, Sb, P) effective against dezincification |
| Alpha-beta brass (C360, C370) | Above 35% Zn | Two-phase (alpha + beta) | Beta phase undergoes preferential dissolution, creating patchy oxide and porous structures |

The beta phase contains approximately 45% zinc and is "chemically active" — no alloying additives can fully protect it during electrochemical processing. In wrought rod, beta phase often appears as stringers that allow corrosion to progress deep into the part. ISO 6509 testing shows dezincification depth ranging from 200 microns in dezincification-resistant alloys (C69300) to 760 microns in highly susceptible alloys (C37700).
Selection guideline: Specify alpha-phase alloys (C260, C270, C69300) for any brass anodizing application. Avoid duplex or high-zinc alloys like C360 or C370.
Once alloy selection is settled, surface condition becomes the next critical variable.
Surface Preparation Quality
Any contamination, deep machining marks, or surface porosity will telegraph through the anodized finish. The Copper Development Association Design Handbook specifies that copper alloy surfaces must be cleaned to remove process oils and oxides before finishing, with alkali cleaning noted as effective.
Since the oxide layer is only 20-500 nanometers thick in research applications (versus aluminum's 5-75 micrometer layers), even minor surface imperfections become visible after anodizing.
Dimensional Tolerance Planning for CNC Machined Parts
The anodized layer adds thickness both above and below the original surface. For threaded features, mating surfaces, and tight-tolerance fits, this dimensional change must be accounted for during the machining stage.
Unlike aluminum anodizing — where Type II follows a 33% buildup / 67% penetration ratio and Type III follows the "50/50 rule" — no equivalent dimensional growth ratio has been documented for brass anodizing. The mixed copper-zinc oxide chemistry and variable Cu:Zn dissolution behavior make a fixed ratio unreliable.
This means dimensional compensation for brass parts needs to be determined empirically — ideally by working with a provider that handles both machining and anodizing, so tolerance adjustments are tested and tracked across both operations rather than estimated in isolation.
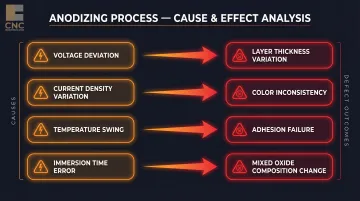
Process Parameter Consistency
Voltage, current density, bath temperature, and immersion time must all be held within defined ranges. Deviations cause:
- Layer thickness variation across the part
- Color inconsistency (especially visible in dyed parts)
- Adhesion failure at parameter extremes
- Mixed oxide composition changes that affect corrosion resistance
Unlike aluminum anodizing (governed by MIL-PRF-8625F with defined parameter windows for Types I, II, and III), brass anodizing lacks standardized specifications. Each shop may develop proprietary parameters, making consistency dependent on internal process control rather than external standards compliance.
Sealing Quality and Post-Treatment Handling
Improper sealing leaves the oxide layer porous and vulnerable to moisture penetration, dye leaching, and corrosion. Rough handling after anodizing but before sealing is complete can cause surface damage that is difficult or impossible to reverse without stripping and reprocessing the part.
Common Misconceptions and When Brass Anodizing Isn't the Right Choice
The Most Common Misconception: Brass Anodizes Like Aluminum
Many assume brass anodizes as reliably as aluminum because both are metals treated electrochemically. In reality, the mixed copper-zinc oxide layer is less stable, and color consistency is harder to guarantee. Teams that plan brass anodizing without testing the specific alloy first frequently encounter patchy or off-color results.
Caswell Plating explicitly states: "Brass cannot be conventionally anodized like aluminum because it does not form a stable, protective oxide layer." Electrochemical oxidation of brass is possible and produces measurable benefits, but it differs fundamentally from standardized aluminum anodizing.
That distinction matters most when selecting a finish for functional applications — which is where alternatives often outperform brass anodizing.
Where Brass Anodizing Is Not the Best Fit
Consider alternatives when:
- Abrasion resistance is the priority — hard chrome (QQ-C-320B) reaches 600-850 HV; electroless nickel hits up to 1000 HV after heat treatment at 340°C
- Opaque, uniform color is required — powder coating delivers consistent coverage without the translucent quality of dyed anodized layers
- The alloy runs high in zinc (above 35% Zn) — electroless nickel or hard chrome are proven alternatives for these compositions
- Standards compliance is non-negotiable — no ASTM, ISO, or MIL-SPEC standard governs brass anodizing; electroless nickel follows ISO 4527, ASTM B733, and MIL-C-26074; hard chrome follows QQ-C-320B
Comparison with Proven Alternatives
| Property | Brass Anodizing | Hard Chrome (QQ-C-320B) | Electroless Nickel (ASTM B733) |
|---|---|---|---|
| Hardness | Not standardized | 600-850 HV | Up to 1000 HV (heat-treated) |
| Salt spray resistance | Not standardized | Depends on nickel underplate | Up to 1000 hours on copper alloys |
| Thickness range | 20-500 nm (research) | Min 0.002 inch (51 micrometers) | 5-50 micrometers |
| Standards | None | QQ-C-320B, ASTM B650 | ISO 4527, ASTM B733, MIL-C-26074 |

For functional corrosion and wear applications on brass substrates, electroless nickel is the better-documented and higher-performing alternative.
Heat Coloring vs. True Anodizing
Heating brass to change its color via oxidation (sometimes called "heat anodizing") is a different process with no electrical current. It produces color but does not deliver the same corrosion resistance or layer stability as true electrochemical anodizing.
Comparative analysis from SG Trading Asia shows:
- Heat coloring: nanometer-scale layers, "minimal to fair" corrosion resistance, "poor to fair" paint adhesion, "poor" wear resistance
- Electrolytic anodizing: thicker durable oxide, "excellent" corrosion resistance, "excellent" adhesion, "good to excellent" wear resistance
If your application requires corrosion protection or wear durability, specify electrolytic anodizing explicitly — heat coloring won't meet those requirements.
Conclusion
Brass can be anodized, and the process delivers real performance and aesthetic benefits. Success, however, depends on alloy selection, precise process control, and upfront tolerance planning, especially for machined components with tight dimensional requirements.
Brass anodizing is not interchangeable with aluminum anodizing: it involves mixed-oxide chemistry, variable Cu:Zn behavior, and no governing MIL-SPEC or ASTM standard. Alpha-phase alloys (up to ~35% zinc) respond consistently; high-zinc alloys produce patchy, unreliable finishes.
That absence of standardization matters. When functional performance and traceability are priorities, documented alternatives like electroless nickel or hard chrome plating are the more defensible choice.
For decorative applications and moderate corrosion resistance on compatible alloys, brass anodizing remains a workable option. Treat it as a custom process that requires alloy-specific validation and documented process parameters — not a drop-in specification borrowed from aluminum finishing workflows.
Frequently Asked Questions
What is an anodized brass finish?
An anodized brass finish is a conversion coating formed electrochemically on brass, creating a bonded oxide layer (CuO, Cu2O, ZnO) that improves hardness and corrosion resistance while enabling decorative color options through dyeing. Unlike plating, the layer grows from the brass substrate itself and will not peel or flake.
What color is anodized brass?
Natural anodized brass retains a warm gold or yellow tone from the underlying copper-zinc alloy. Dyeing during the anodizing process (while the oxide layer is still porous) can achieve black, blue, green, red, brown, and custom colors while maintaining a metallic sheen beneath the transparent oxide.
Does an anodized brass finish scratch off?
The oxide layer will not peel or flake, but it can wear down in high-abrasion environments over time. The layer is harder than base brass yet more brittle, so sustained mechanical friction is its main vulnerability.
Can you anodize aluminum to look like brass?
Aluminum can be dyed or colored post-anodizing to approximate a warm gold or brass tone, though it will not replicate the natural warmth and depth of actual brass. This is a common design workaround when weight reduction matters, as aluminum is significantly lighter than brass.
Does anodized aluminum react with brass?
Yes — galvanic corrosion risk exists when anodized aluminum contacts brass in the presence of moisture. Aluminum is more anodic than brass and will corrode preferentially, so isolate the two metals with non-conductive coatings, greases, or physical barriers in wet environments.
What is the 7-20 rule for anodizing?
No authoritative source documents a "7-20 rule" for anodizing. Documented dimensional growth ratios apply to aluminum only — Type II follows 33% buildup / 67% penetration, and Type III follows the "50/50 rule" — with no equivalent established for brass due to its variable mixed-oxide chemistry.


