
Introduction
Aluminum ranks among the most widely used metals in modern manufacturing and CNC machining—yet the same 6061-T6 alloy can perform dramatically differently based on one variable: surface finish. A bracket machined from identical stock can fail within months in a coastal environment if left in mill finish, yet thrive for years when clear anodized.
The choice between mill finish and clear anodized aluminum directly impacts corrosion resistance, surface durability, dimensional tolerances on precision parts, maintenance cycles, and total project cost. For decision-makers in manufacturing, this isn't just an aesthetic preference—it's a critical design specification that influences part longevity and operational reliability.
What follows breaks down both finishes side by side—covering performance data, dimensional implications, and a practical framework for choosing the right surface treatment for your next component order.
TL;DR
- Mill finish aluminum is raw, untreated metal straight from the extrusion die—lowest cost, accepts secondary coatings, ideal for dry or indoor applications
- Clear anodized aluminum features an engineered oxide layer grown through electrochemical processing—delivers significantly greater corrosion and abrasion resistance with a uniform satin-silver finish
- Oxide layer thickness is the core difference: mill finish has a natural 2-4 nm layer; clear anodized builds 5-25 micrometers—up to 5,000× thicker
- Choose mill finish for budget-sensitive projects, hidden structural components, or parts receiving paint or powder coat
- Clear anodized is the right call for parts exposed to moisture, corrosive environments, or visible applications where surface quality matters
Mill Finish Aluminum vs. Clear Anodized: Quick Comparison
Cost
Mill finish: Lower unit cost—no secondary finishing process required, making it the economical baseline for aluminum components.
Clear anodized: Higher cost due to additional electrochemical processing. Industry sources indicate anodizing infrastructure can add 25% or more to base capital costs, with per-part pricing shaped by throughput, batch size, and process control requirements.
Surface Protection (Oxide Layer)
Mill finish: Natural aluminum oxide layer approximately 2-4 nanometers thick, chemically bonded but offering minimal protection depth.
Clear anodized: Engineered anodic aluminum oxide (AAO) layer typically ranging from 5-25 micrometers thick (Type II per MIL-A-8625F), with depth controlled by process parameters. That translates to a protection layer thousands of times thicker than mill finish.
Appearance
Mill finish: Dull, matte gray-silver appearance with visible die lines, extrusion marks, and surface inconsistencies characteristic of the raw manufacturing process.
Clear anodized: Uniform satin-silver sheen with smooth, polished appearance—extrusion imperfections are removed during chemical pre-treatment, resulting in consistent visual quality across production runs.
Corrosion and Abrasion Resistance
Mill finish: Passively resistant under normal indoor conditions but vulnerable in humid, salty, or chemically aggressive environments. Atmospheric corrosion rates range from 0.03 micrometers/year in rural settings to 13 micrometers/year in polluted environments.
Clear anodized: Far more resistant to corrosion and surface wear. Type II clear anodize must pass 336 hours of salt spray testing per MIL-A-8625; architectural Class I anodize passes 3,000 hours per AAMA 611 standards. The anodized layer measures 250-370 Kgf/mm² hardness—harder than untreated base aluminum.

Best Fit Scenario
Mill finish: Indoor structural components, prototypes, parts destined for paint or powder coat, cost-driven production runs where environmental exposure is minimal.
Clear anodized: Exterior-facing parts, visible consumer or architectural components, applications in coastal or high-humidity environments, precision parts requiring long-term dimensional stability.
What is Mill Finish Aluminum?
Mill finish aluminum is aluminum delivered directly from the extrusion die or rolling mill without any post-process surface treatment. The term "mill finish" indicates the product remains in its raw, as-produced state — the surface characteristics from manufacturing are what you get.
When exposed to air, bare aluminum spontaneously forms a thin aluminum oxide (Al₂O₃) layer approximately 2-4.3 nanometers thick. This naturally occurring oxide chemically bonds to the aluminum surface and provides passive corrosion resistance in environments with pH roughly between 4 and 9 (source). Below pH 4 or above pH 9, corrosion accelerates significantly.
Visible characteristics include:
- Dull, matte gray-silver color
- Visible die marks from the extrusion process
- Surface streaks or minor discoloration
- Uneven surface finish compared to treated aluminum
Core advantages for manufacturing operations:
- Costs less than any treated alternative — no finishing step means no added budget
- Retains full recyclability value without coating contamination
- Maintains 61% IACS conductivity, making it suitable for electrical contact (unlike anodized coatings, which act as insulators)
- Works as a ready substrate for paint, powder coat, or anodizing without compatibility issues
Key limitations manufacturers must account for:
- Uneven surface finish unsuitable for visible applications
- Minimal abrasion resistance—surface scratches easily
- Corrosion susceptible in moist, salty, or acidic environments
- Natural oxide layer can be mechanically damaged through handling
Use Cases of Mill Finish Aluminum
Mill finish suits workflows where aluminum stays in controlled environments or moves into a secondary finishing process. It's the natural starting point for structural components, interior assemblies, frames, brackets, and parts headed downstream for coating.
Industries where mill finish dominates:
- Construction framing and building infrastructure
- HVAC ducting and ventilation systems
- Electrical enclosures for interior applications
- Aluminum packaging (foil, beverage cans)
- Industrial machinery frames and structural supports
- Rapid prototyping environments prioritizing speed and budget over aesthetics
What is Clear Anodized Aluminum?
Clear anodized aluminum has undergone an electrochemical process that grows a thick, controlled aluminum oxide layer on the metal surface. "Clear" simply means no dye or pigment was added — the result is a satin-silver finish that preserves aluminum's natural metallic appearance while measurably improving hardness, corrosion resistance, and wear life.
The Anodizing Process
Anodizing runs in five steps:
- Pre-treatment/etching: Removes surface imperfections, oils, and the natural oxide layer, creating a clean substrate
- Electrolyte bath immersion: Parts submerge in a sulfuric acid solution that serves as the electrolyte medium
- Electrical current application: Direct current flows through the bath, with the aluminum part serving as the anode
- Controlled oxide growth: The anodic aluminum oxide layer grows simultaneously into and out of the aluminum substrate
- Pore sealing: Hot water or nickel acetate treatment closes the porous structure, completing corrosion protection

Structural Properties of the Anodic Layer
The anodized layer features a self-organized honeycomb-like structure with uniform parallel pores ranging from 5 nanometers to several hundred nanometers in diameter. This porous microstructure allows dye absorption for colored finishes (though clear anodize skips this step) and requires sealing to achieve maximum corrosion protection.
According to MIL-A-8625F specifications, Type II anodize thickness ranges from 0.0002" to 0.001" (approximately 5-25 micrometers). Layer thickness can be precisely controlled through bath temperature, acid concentration, current density, and processing duration.
Key performance advantages:
- Sealed anodic coatings resist atmospheric and salt-water attack, making them reliable in coastal and high-humidity environments
- Surface hardness reaches 250-370 Kgf/mm² — far above base aluminum — giving the finish genuine scratch and abrasion resistance
- The uniform satin-silver finish eliminates die marks and surface inconsistencies without added coatings
- The oxide layer is part of the metal structure, not applied on top — it cannot chip or peel like paint
Key limitations for precision manufacturing:
- Electrochemical processing adds cost compared to untreated mill finish
- The oxide layer acts as an electrical insulator, reducing conductivity — a real concern for grounding or EMI applications
- Slight batch-to-batch color variation can occur depending on alloy composition and process parameters
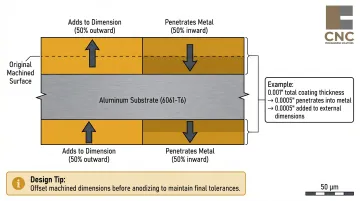
- Approximately 50% of the coating penetrates into the substrate and 50% grows outward — critical for tight-tolerance CNC parts, where designers must offset dimensions to account for this outward growth
Use Cases of Clear Anodized Aluminum
Given those tradeoffs, clear anodized aluminum works best when the anodized layer is the final finish — not a primer for something else. It earns its place on parts that face environmental stress, need a durable appearance, or must meet specific surface standards.
Industries where clear anodized aluminum dominates:
- Aerospace: Structural components and aircraft skins governed by MIL-A-8625 specifications
- Architecture: Window frames, curtain walls, and facades meeting AAMA 611 Class I standards (minimum 0.7 mil thickness, 3,000-hour salt spray resistance)
- Consumer electronics: Laptop housings, smartphone frames, and tablet enclosures requiring scratch resistance and premium aesthetics
- Medical devices: Equipment casings that withstand repeated sterilization cycles
- Marine hardware: Components exposed to salt-laden coastal environments
- Automotive trim: Exterior and interior decorative elements requiring durability
Mill Finish Aluminum vs. Clear Anodized: Which is Better for Your Project?
Neither finish is universally superior—the right choice depends on your specific application requirements. Evaluate these key decision factors:
Environmental Exposure
Choose mill finish if your parts operate indoors in controlled, dry environments with minimal exposure to moisture, salt, or corrosive chemicals. Mill finish performs adequately within pH 4-9 ranges under normal atmospheric conditions.
Choose clear anodized if parts face outdoor exposure, coastal/marine environments, high humidity, or contact with corrosive substances. The oxide layer provides measurably better protection—architectural Class I anodize passes 3,000 hours of accelerated salt spray testing compared to no standardized test requirement for mill finish.
Aesthetic Requirements
Choose mill finish for hidden structural components, internal assemblies, or parts where appearance doesn't impact product value. The dull, inconsistent surface finish is functionally adequate but visually unrefined.
Choose clear anodized for consumer-facing parts, architectural applications, or any component where surface quality contributes to perceived product value. The uniform satin-silver finish eliminates extrusion marks and provides consistent visual quality.
Budget and Production Volume
Cost sensitivity often drives this decision. Consider these trade-offs:
- Mill finish: No finishing process means lower per-unit cost and faster turnaround — the right call when environmental and aesthetic demands are minimal
- Clear anodized: Adds upfront cost but the extended service life, reduced maintenance, and fewer field failures typically justify the investment in demanding applications
Secondary Coating Plans
Choose mill finish if parts will receive paint, powder coat, or other secondary finishes. Mill finish serves as an ideal substrate without adding unnecessary processing steps or cost.
Choose clear anodized if the anodized layer will serve as the final finish with no additional coating required.
Dimensional Tolerance Sensitivity
Critical consideration for precision-machined parts: Anodizing adds measurable thickness to every surface. According to MIL-A-8625F, approximately 50% of the anodic layer penetrates into the metal and 50% grows outward—meaning a 0.001" (25 micrometer) anodic coating adds 0.0005" to external dimensions.
For tight-tolerance CNC-machined aluminum parts, designers must offset machined dimensions before anodizing to maintain final dimensional specifications. Mill finish does not alter part geometry, making it the lower-risk option for critical tolerances when environmental protection isn't required.
Finish Selection in CNC-Machined Aluminum Parts: A Real-World Scenario
Consider a manufacturer producing aluminum enclosures for industrial electronics who initially specified mill finish parts to control costs. Over 18 months of field deployment, units installed in humid or coastal locations showed visible surface corrosion, while customer complaints about appearance quality increased—prompting a finish strategy re-evaluation.
The solution involved strategic finish selection based on component visibility and exposure:
- External surfaces: Switched to clear anodized finish for all customer-visible faces where corrosion resistance and aesthetics directly impact perceived product quality
- Internal structural brackets: Retained mill finish for hidden components where appearance is irrelevant and cost savings are meaningful
- Result: Extended service life, reduced maintenance calls, and improved customer satisfaction
Industrial PC manufacturers report similar outcomes when deploying anodized enclosures in factory automation, outdoor, and marine environments. Common benefits cited include extended enclosure lifespan, fewer corrosion-related failures, and lower total cost of ownership despite higher upfront material cost.
For manufacturers navigating these decisions, working with a shop that handles both machining and finishing under one roof simplifies the process. CNC Programming Solutions provides anodizing and other finishing services as part of its precision machining workflow, so finish specifications carry through from programming to final part—no separate vendor coordination required. Reach the team at 405-714-3714 or cncsolutions22@gmail.com to discuss your aluminum finishing requirements.
Conclusion
Mill finish aluminum makes sense when cost control, machinability, and post-treatment flexibility are priorities—particularly for indoor structural components or parts headed for secondary coating. Clear anodized aluminum earns its place when performance, longevity, and appearance must hold up under demanding environmental conditions or visible applications. Your operational requirements drive the decision, not a universal ranking.
Getting this wrong carries real costs: corrosion failures, accelerated maintenance cycles, field rework, and customer dissatisfaction. Clear anodizing is worth the upfront investment when service conditions demand it. Mill finish remains the efficient, cost-conscious default when those conditions don't apply.
Frequently Asked Questions
What is the difference between mill finish aluminum and anodized aluminum?
Mill finish is aluminum in its raw, uncoated state with only a naturally occurring thin oxide layer (~2-4 nanometers), while anodized aluminum has undergone an electrochemical process to grow a much thicker protective oxide layer (5-25 micrometers for Type II). That thickness difference translates directly to improved corrosion resistance and surface hardness — at a higher per-part cost.
What does mill finish aluminum look like?
Mill finish aluminum has a dull, matte gray-silver appearance with visible extrusion die lines, surface streaks, and minor inconsistencies from the manufacturing process. It lacks the uniform, polished sheen characteristic of anodized or otherwise treated aluminum surfaces.
Will mill finish aluminum rust?
Aluminum cannot rust—rust is specifically iron oxide (Fe₂O₃) and occurs only in iron-containing metals. However, mill finish aluminum does corrode when exposed to moisture, salt, or corrosive chemicals, forming a white powdery aluminum oxide residue. Unlike iron rust, aluminum's natural oxide is self-passivating and provides limited protection within pH 4-9 ranges.
Can mill finish aluminum be painted?
Yes, but proper surface preparation — cleaning, etching, and priming — is required for adhesion. Without it, paint and powder coat are prone to peeling. Mill finish actually accepts secondary finishes more readily than anodized aluminum once the surface is correctly prepped.
What can you not use on anodized aluminum?
Avoid highly alkaline cleaners (bleach-based or caustic soda products) and strong acids, which can damage the anodic oxide layer. The Aluminum Anodizers Council recommends mild soap or detergents with pH 6-8 only. Abrasive scrubbing pads and steel wool can breach the anodized surface—use soft cloths instead.
Is mill finish aluminum better than clear anodized aluminum?
Neither finish suits every application. Mill finish wins on cost and downstream coating flexibility; clear anodized wins on corrosion resistance, surface hardness, and appearance consistency. Match the choice to your environmental exposure, aesthetic requirements, and budget.


